Citrus gall wasp in Southern Australia
Summary
Citrus gall wasp (Figure 1), Bruchophagus fellis, is an Australian native insect pest whose natural host is the Australian finger lime (Citrus australasica).
CGW has traditionally been a pest of citrus trees in Queensland and mid to north NSW. It has recently established in the southern citrus production regions of Sunraysia, Riverland and the Riverina, where Australian orange production is concentrated.

Fast FAQ
Q: When do adult Citrus gall wasp appear in the orchard?
A: Citrus Gall Wasp (CGW) adults are most abundant from late October to mid November and are present for about four weeks. Most adult wasps stay near their tree of emergence. An online tool predicting adult wasp emergence and egg hatch has been developed and is available from the NSW DPI website (www.dpi.nsw.gov.au/agriculture/horticulture/citrus).
Q: What chemical control options are available?
A: Two soil-applied systemic insecticides target larvae and provide effective control:
Clothianidin (Samurai® Systemic Insecticide) is applied via micro-irrigation after flowering has finished. The product label outlines important critical use comments for applying the product to citrus.
Imidacloprid (Confidor® Guard Soil Insecticide) has a citrus gall wasp suppression only claim on the label. Imidacloprid (Confidor® Guard Soil Insecticide) is also applied through the irrigation system and critical use comments are stated on the product label.
Q: Are there any non-chemical control options?
A: Mechanical pruning or skirting is an important cultural control measure for orchards with heavy citrus gall wasp infestations.
Q: Do I need to destroy galls that have been pruned out of trees?
CGW adults can continue to emerge from galls after removal from the tree. It is advisable to move the prunings into the row centre and mulch them if pruning is done within 8 weeks of the expected emergence date.
Q: Will CGW be controlled by beneficial insects?
A: Parasitic wasps are present in all southern growing regions, but are not in sufficient numbers to provide satisfactory control. Periodic releases of parasitic wasps will boost numbers and improve biological control. Soil-applied systemic insecticides will kill citrus gall wasp parasitoids.
Q: Do I need to apply controls every year?
A: No. Monitoring is essential as low levels of CGW infestation may not require chemical intervention. Applying systemic soil chemical controls every season is not recommended, as it can lead to other pest problems and increase the risk of insecticide resistance.
Life cycle
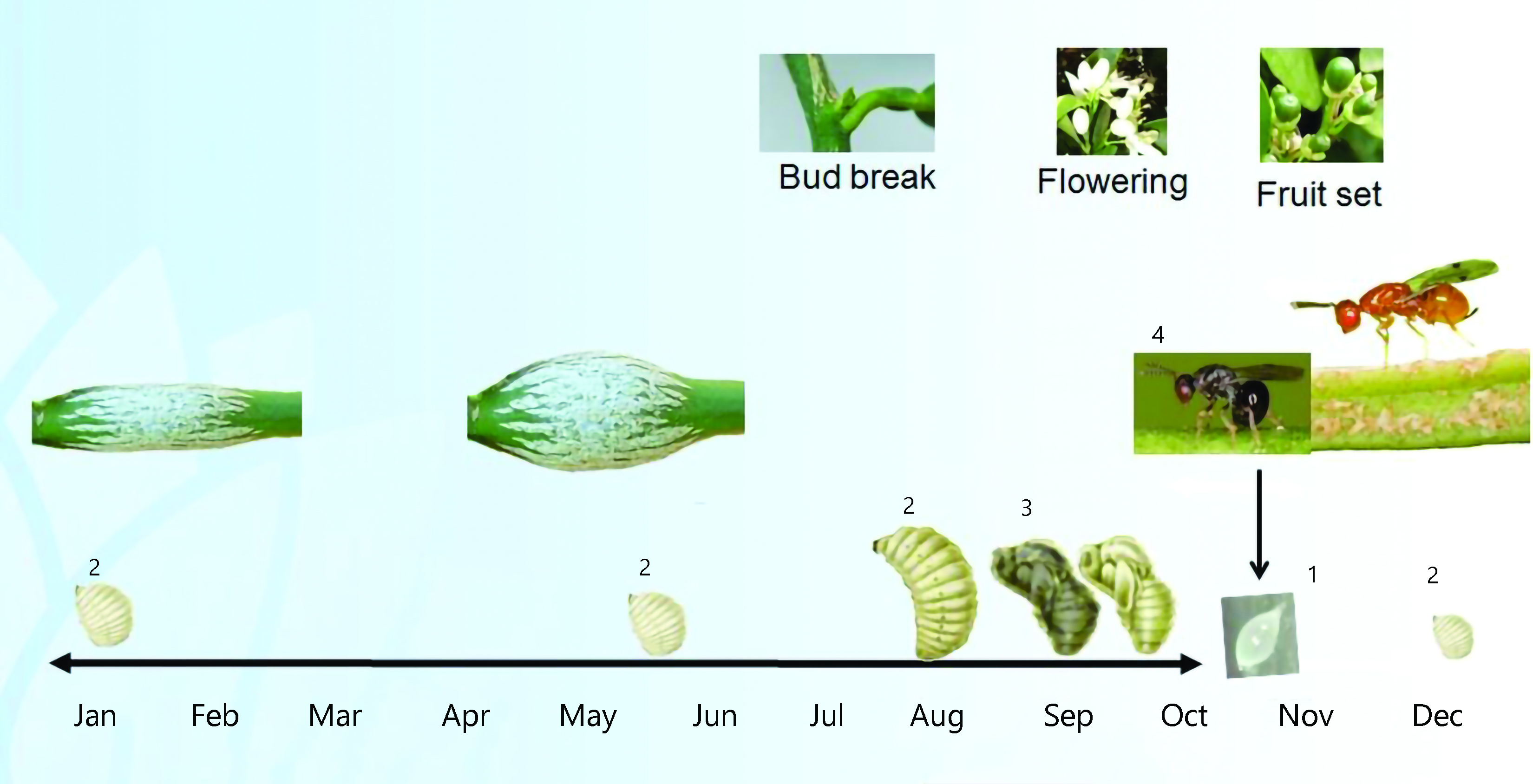
The CGW life cycle consists of an egg stage, four larval stages, a pre-pupa stage, a pupa stage and an adult stage (Figure 2). Except for the adults, all stages are completed inside the galls (Figure 3). The wasp has a single generation per year.
Adult CGW emerge from galls in spring (Figure 4). This timing is influenced by temperature and closely associated with the appearance of the spring growth flush. In the southern regions, adult wasps emerge between mid October to mid December and are most abundant from late October to mid November. Peak emergence occurs after an accumulation of 573 degree days above 11 °C from 1 June.
Adult CGW do not fly very far; research shows that 95% stay in the tree where they emerged. However, they can be transported over long distances on prevailing winds.
Adult CGW mate and lay eggs on the same day of emergence. Most eggs are laid within three days. Adult CGW live for 3–14 days depending on the temperature. Each female can lay about 100 eggs. Eggs are mostly laid under the bark of young spring shoots. Larvae hatch in 2–4 weeks.
Long-distance movement of CGW around Australia occurs mainly through the distribution of infected citrus nursery trees. It is recommended that new trees should be thoroughly checked for the presence of galls. If galls are found, advise the nursery. Isolated galls can be cut out or painted over with a specialised sticky paste (e.g. Tanglefoot®) to trap the wasps as they emerge.


Damage
Heavily galled trees lose vigour, which can result in reduced fruit size (Figure 5) and yield, and in extreme cases, branch dieback. CGW infests young flush growth in spring, causing woody galls to form around the developing larvae. A CGW gall houses many wasps each in a separate cell. All varieties can be attacked but lemons, grapefruit, and some rootstocks (e.g. Trifoliata) are usually more heavily infested.

Monitoring
Monitoring of CGW is best done from autumn onwards when galls are easily seen. Low level infestations are very difficult to detect since galls are small (Figure 6) and only a few are present on a tree. Galls tend to be more abundant in the lower canopy close to the skirt line. A good monitoring technique is to check underneath the canopy and look for shoots growing at right angles from the main branch (Figure 7). Galls are commonly seen on rootstock suckers and these can then be targeted for monitoring.
The severity of CGW within a block can be estimated using the branch unit method. This monitoring technique uses a 2–3 year old branch (inclusive of all shoots on a 30 cm long branch) as the basic sampling unit (Figure 8). Thirty branch units should be assessed for up to a five hectare block. When sampling branches, ensure different heights and aspects of the canopy are included. Sampled trees should be representative of the block. Each branch unit is assessed by recording the percentage of branches with galls present, as well as the size and number of these galls. A sample recording sheet is given in Table 1.



Table 1. An example of a Citrus gall wasp monitoring data sheet.
| Branch | < 3 cm | 3–5 cm | > 5 cm |
|---|---|---|---|
| 1 | 2 | 0 | 0 |
| 2 | 0 | 1 | 1 |
| 3 | 0 | 0 | 0 |
Action thresholds
There are no definitive action thresholds established for CGW. The decision to apply chemical treatments should take into consideration the severity of infection based on monitoring results and the individual farm situation (Table 2).
Table 2. Citrus gall wasp monitoring interpretation guide.
Rating | % branches with galls | Galls per branch | Comments and options |
|---|---|---|---|
Low | < 10% | < 0.2 | Galls are generally less than 3 cm. Consider treatment if it is a new incursion on a previously uninfested orchard. Low priority for treatment in orchards with existing low levels. |
Moderate | 10–50% | < 2 | Increasing numbers of small galls but still no or very few galls greater than 5 cm and no immediate economic impact. Populations would be anticipated to increase rapidly within the next 2–3 seasons. As a guide, one in three branches infested with galls (30%) might be a medium priority and a starting point to consider insecticide intervention as part of area-wide management. |
Significant | > 50% | > 2 | Numerous galls above 5 cm are present. Economic impact will be evident within a few seasons. High priority area for insecticide intervention. |
Management
Cultural practices
Adult CGW are only present in the orchard in spring and early summer and predominately lay eggs into spring flush. In orchards where CGW is restricted to isolated trees, gall-bearing branches should be removed and destroyed if within six weeks of emergence.
Galls are often more abundant on rootstock suckers. Desuckering trees following CGW emergence will allow the rootstock suckers to act as a trapping site. This will assist in slowing the establishment of CGW in new incursion areas.
In heavily infested orchards, trees can be pruned (i.e. heavy hedging) to rejuvenate the canopy. CGW can still emerge from cut galls if left in the shade for less than six weeks. Mulching these prunings after hedging will help to dry out galls and kill the wasp before emergence. Be aware that heavy pruning will encourage vigorous regrowth which will be highly attractive to remaining CGW adults when they emerge and this regrowth needs protecting.
Galls appear to be more abundant in the lower parts of the tree canopy, which could be a result of shading and a more humid micro-climate. CGW also favour under-tree sprinklers, rather than the drier conditions of drip irrigation. Skirting trees after adult CGW emergence (over summer) can be useful in reducing establishment in some situations.
Involving neighbours in an area-wide management approach will provide the best results.
Biological control
Two parasitic wasps, Megastigmus brevivalvus and M. trisulcus, are important natural enemies of CGW (Figure 9, Figure 10 and Figure 11). They are similarly sized to CGW but are honey-coloured and can be easily distinguished from the black CGW adults.
Parasitic wasps insert their eggs directly into CGW eggs. After hatching, the larvae feed and develop inside CGW larvae, eventually killing the CGW. Up to 90% of gall wasp larvae can be parasitised. Both wasp species have established in the southern citrus regions with M. brevivalvus the more abundant species.

To date, parasitic wasp populations in the southern citrus regions have only been recorded at low levels (i.e. less than 10%); insufficient numbers to satisfactorily control CGW populations. Parasitism levels can be checked by storing some galls in clear containers with perforated lids.
Parasitic wasp populations will gradually build up over many years, reducing the dependence on chemical control. Natural populations of parasitic wasps alone are not expected to provide sustained biological control of CGW. This is because their populations fluctuate with CGW populations and consequently vary greatly from season to season. Periodic releases of parasitic wasps are required for improved biological control.


Parasitic wasps suffer high mortality during heatwaves. Light irrigation provides a drinking water source to the parasitic wasps and will help increase their survival.
Parasitic wasps can be introduced or boosted by collecting wasps from known hotspots. This can be achieved by collecting galls at the beginning of parasitic wasp emergence (towards the end of the CGW emergence period) and transporting them to the new release site. Galls should be placed in the shaded part of the tree as soon as possible after collection in open containers. Repeated releases for 3–5 years may be needed to establish parasitic wasps.
The feasibility of commercial mass rearing of parasitic wasps is being investigated.
Chemical control
Control strategies need to reduce the emergence of adult CGW by at least 90%. This level of control is required because female CGW can lay many eggs, and even if a moderate proportion survives, they can quickly re-establish numbers. All chemical controls currently available for CGW are considered disruptive to biological control and integrated pest management (IPM). Chemical controls should only be applied if monitoring indicates action is required (Table 2). For export fruit, check the compliance of all chemicals with overseas maximum residue limits.
Contact foliar insecticides
No contact foliar insecticides are registered to control adult CGW. Adult wasps emerge over a 4 week period and current insecticides do not have adequate residual activity to provide effective control. Eggs will be laid prior to insecticide application and also after the chemical’s residual activity has ended. Repeatedly applying contact insecticides is highly disruptive to citrus IPM.
Systemic and translaminar insecticides
Systemic and translaminar chemicals target the larvae that hatch from the eggs. Eggs are not affected. Female CGW lay most eggs within three days of emergence and eggs hatch 3–4 weeks later.
Two Group 4A neonicotinoid soil-applied systemic insecticides, Clothianidin (Samurai® Systemic Insecticide) and Imidacloprid (Confidor® Guard Soil Insecticide), are available for CGW control and suppression. Apply systemic insecticides after flowering and in accordance with permit and label recommendations. One or two years of consecutive applications should bring levels down to where no further action is required for a number of years. If controls are not effective, the application method should be reassessed. Continuous use of soil-applied systemic insecticides is not recommended and can cause other pest problems.
Integrated strategies
Controlling CGW on the farm should involve a number of strategies. Some factors to consider include:
- monitoring will help identify levels of infestation.
- prioritising treatment areas and developing a plan which leaves some areas untreated (i.e. low priority) to provide a repository for beneficial insects.
- occasional and strategic chemical applications (e.g. two years of chemical application followed by two or more years of no treatment depending on monitoring) will reduce the risk of chemical resistance and encourage biological control.
- encouraging neighbours to control their CGW populations and annually discuss planned strategies with each other.
- multiple strategies might be needed to control severe infestations (e.g. pruning and systemic controls).
Citrus gall wasp project report
Project final reports are free to Australian horticulture levy payers, registered members and Industry Representative Bodies. Load the Hort Innovation website and complete the required information on the final report order form: https://horticulture.com.au/resources/final-report-order-form/.
References
Hardy S and Creek A. 2009. Final Report – Citrus Gall Wasp Technical Forum 5th May 2009, NSW Department of Primary Industries, Orange.
Mo J, Creek A, Munro S and Stevens MM. 2014a. Managing citrus gall wasp in southern citrus regions. Final Report, HAL CT10021, Horticulture, Australia Ltd, Sydney.
Mo J and Stevens MM. 2014b. Emergence patterns of adult citrus gall wasp, Bruchophagus fellis (Hymenoptera: Eurytomidae), and its key parasitoids in southern Australia. Journal of Asia-Pacific Entomology 17: 311–317.
Noble, NS. 1936. The citrus gall wasp. Science Bulletin No. 53, Department of Agriculture NSW, Sydney.
Smith, D, Beattie, GAC and Broadley R (eds) 1997, Citrus pests and their natural enemies – integrated pest management in Australia, Queensland Department of Primary Industries, Brisbane.
Authors
Jianhua Mo, Entomologist
Steven Falivene, Citrus Development Officer
Andrew Creek, Citrus Development Officer
Acknowledgements
This revised Primefact is an output of the Development of national strategies to manage citrus gall wasp (CT15006) project funded by Horticulture Innovation, using the citrus research and development levy, contributions from the Australian Government and NSW Department of Primary Industries. Hort Innovation is the grower owned, not-for-profit research and development corporation for Australian horticulture.

Craig Swanbury, Naomi Galbraith and Narrelle Borgmeyer (Fruit Doctors) are acknowledged for their participation in the project and contribution to this Primefact.
This Primefact is a revision of a previous version by Sandra Hardy (formerly NSW DPI) and Andrew Creek.
Dan Papacek (Bugs for Bugs) and Dr Andrew Beattie (University of Western Sydney) are acknowledged for their input. Richard and Judy Bertalli (Sunraysia citrus growers) are acknowledged for their participation and knowledge shared during NSW DPI and MVCB trials conducted in 2010 and their ongoing collaboration. Mary Cannard is acknowledged for her facilitation of the MVCB gall wasp trials. Shane Smythe (Sunraysia citrus grower) is acknowledged for his collaboration. Robert Pandolfo, Garry Pandolfo and Kevin Cock are acknowledged for their participation in NSW DPI trials conducted in 2016/17.