Assessing and managing resistance to postharvest fungicides: Mar 2018
Fungicide resistance management
This article contains a summary of the fungicide resistance survey that was completed in the 2017 season. The results show there is room for improvement in the hygiene and management of fungicide resistance in Australian packinghouses. This project is a contribution from the Australian Citrus Postharvest Science Program (CT15010).
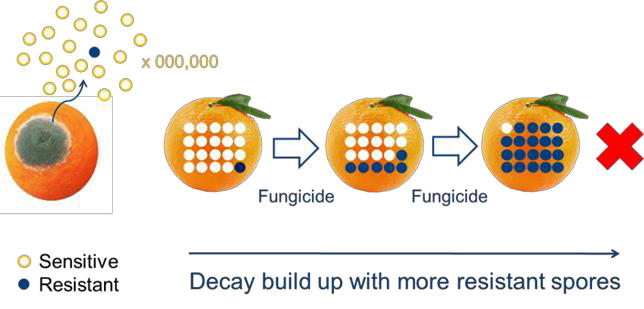
Using postharvest fungicides is an important method to control the development of decay during handling and marketing. However postharvest fungicides can sometimes fail to work due to the development of resistance by the decay fungi to the fungicide. Fungicide resistance is a serious and important postharvest problem which needs to be actively managed in the packing shed to minimise any potential losses.
Fungicide resistance can occur in sheds and cool rooms with poor hygiene and the ongoing use of the same postharvest fungicide. Resistance arises when a single decay fungi spore that is resistant to a fungicide multiplies. The continued selection of these resistant spores can happen when the same fungicide is used and these spores can multiply and full resistance develops.
2017 Survey to assess the presence of decay-causing fungi
During the 2017 packing season, NSW DPI conducted a survey of packing shed hygiene and resistance to postharvest fungicides across Australia as part of the Hort Innovation Citrus Postharvest Program (CT15010).
Agar petri dishes with different fungicides were exposed in sheds during the main packing season, to estimate the levels of fungicide resistance present. The plates were put into the start of the packing line, the end of the packing line, and in the cool room. In the major survey, the postharvest fungicides used were; thiabendazole (TBZ) (Vorlon® or Tecto®) and fludioxonil (Scholar®), whilst in the later stages of the survey, imazalil (Magnate® or Fungaflor®) was added. If decay spores in the air of the packing shed landed on the plates with fungicide and then grew, then there was some technical resistance to that fungicide.
Shed by shed hygiene comparison
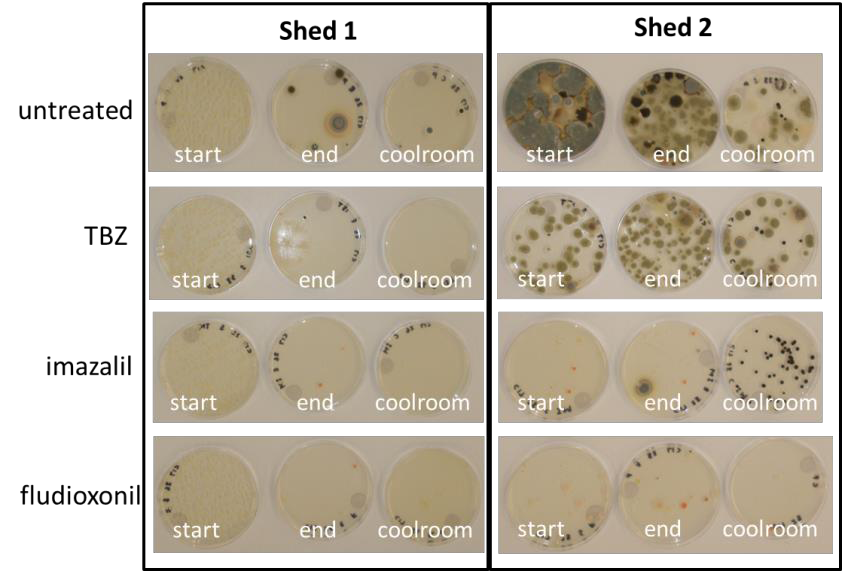
Results show that there are large differences between sheds. The plates that were placed inside Shed 1 had very few fungi spores, even on the untreated plates, which indicated that this shed had good hygiene practices with very low numbers of decay causing fungi. In addition, there was no evidence of any fungicide resistance, with no fungi growing on the plates with fungicide. This is a very good result for this grower and shows the effects of good sanitation and fungicide use.
In contrast, the results of Shed 2 showed relatively high levels of decay-causing fungi spores in the untreated plates indicating relatively poor shed hygiene. In addition, some technical resistance to TBZ and imazalil fungicides was detected, particularly against TBZ. The detection of technical resistance is a concern and management of shed hygiene and resistance is required.
The results of all the different packing shed around Australia showed there was a high level of variability in the management of hygiene and levels of technical resistance between the different packing sheds. The majority of the survey was conducted with TBZ and fludioxonil.
The results showed very little evidence of resistance to fludioxonil, as this fungicide is not widely used by industry. However there were some sheds which had significant technical resistance to TBZ. The management to minimise fungicide resistance requires a whole-of-system approach, starting from harvest through to packing and storage.
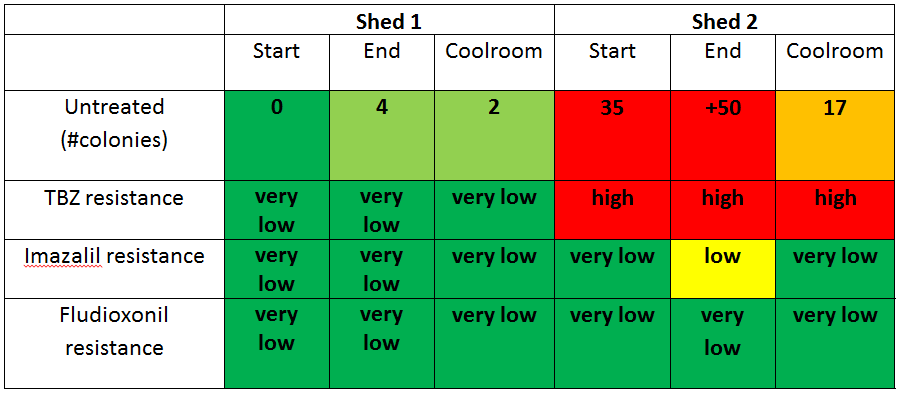
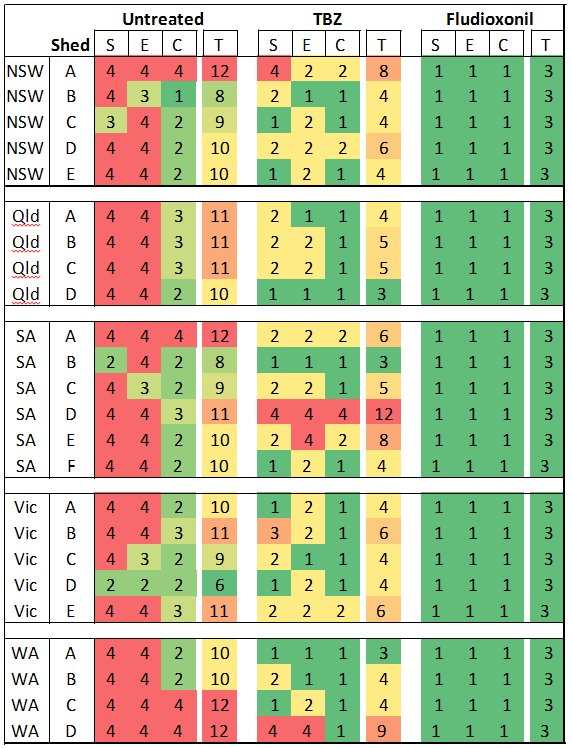
S = Start of the line, E = End of the line, C = Coolroom, T = Total score for all different shed locations (S + E + C). Untreated (control) plates measured hygiene where a score of 1 =very low spore levels, 2 = low spore levels, 3 = moderate spore levels, 4 = high spore levels. For TBZ and fludioxonil amended plates a score of 1 = very low resistance detected, 2 = low levels of resistance detected, 3 = moderate levels of resistance detected, and 4 = high levels of resistance detected.
Reducing resistance
Optimise fruit health
It is good postharvest practice to minimise physical damage to the fruit during harvest and handling. Handle all fruit carefully, as most postharvest decay fungi can only infect through wounds in the peel. Therefore, reducing injury will reduce the incidence of decay.
The ‘Australian Citrus Harvest Handbook’ was released this year and has excellent guidelines on the correct harvest and handling of citrus. In addition, limiting the storage of the fruit and keeping fruit at the optimal storage temperatures reduces the ageing of the fruit and therefore its susceptibility to decay. These factors are not only good postharvest practices, but they also reduce the risk of fungicide resistance.
Use best hygiene practices
Good shed hygiene is crucial to minimise the risk of fungicide resistance. Lowering the population of decay-causing spores in the shed, cool room and on the fruit is key to a successful management program. Fewer fungi spores in the shed reduce risks in the development of resistance.
This includes the regular sanitation of equipment, cool rooms and packing line by washing down (or using fogging technology) to sanitise them. It is absolutely critical to removal all culled and especially decaying fruit from the shed and dispose of this fruit well away from the packing shed. A single rotten fruit in the shed or receival area is a prime source of spores that can inoculate healthy fruit and the entire shed. It is also ideal to have a separate area of receivals of fruit from the orchard to the packing and re-packing lines. Good postharvest hygiene is key to managing resistance. In addition to fungicides, sanitisers should be used to wash and clean the fruit surface.
Optimise fungicide use
As described in the Australian Citrus News (pages 12-14, Spring 2017), fungicides are classified into different groups, depending on the way they work. It is important to understand the way each fungicide works to develop strategies to minimise the development of resistance.
In summary, TBZ is classified as Group 1; imazalil is classified as Group 3, imazalil and pyrimthanil is Group 3 and 9, fludioxonil is Group 12 and guazatine is Group M7. To reduce the risk of fungicide resistance, it is important to limit the total number of fungicide applications of any one class, ideally to one per fruit lot. The continued use of the same fungicide class can cause selection pressures for that chemical resulting in resistance.
Another important management practice to reduce the risk of resistance is to use rotations and mixtures or pre-mixtures whenever possible before resistance selection occurs. This makes it difficult for the fungus to develop resistance as there are different modes of action of each of the mixtures against the fungus. Strictly use the label rate for each fungicide. Lower rates of fungicide can select for less sensitive spores in the population.
Optimise fungicide efficacy
It is important not only to use the correct fungicide but also to use it correctly, as fungicide coverage determines the efficacy of the treatment and minimises the chances of decay spores surviving following treatment. The use of sanitisers and alkaline salts in the packing line to kill pathogens are also a good way to reduce the risk of fungicide resistance. Sanitisers have a broad method of killing fungi, and resistance to these chemicals is unlikely.
Monitor fungicide resistance
It is important to routinely monitor pathogen populations for their sensitivity to postharvest fungicides. The early detection of potential resistance issues increases the chance that its development can be managed and stopped. Routine hygiene monitoring can also help with knowing that your sanitation and cleaning programs are working.
This article was originally published in the Packer Newsletter which is a contribution from the Australian Citrus Postharvest Science Program (CT15010) funded by Horticulture Innovation and NSW Department of Primary Industries. Levies from Australian citrus growers are managed by Horticulture Innovation and contributed to funding this project. The Australian Government provides matched funding for all Horticulture Innovation’s research and development activities.
For accessible version of images used in this article contact:
John Golding
NSW DPI
E: john.golding@dpi.nsw.gov.au P: 02 4348 1926